Laccase Enzyme Production for Industrial Buyers
Buyer guide to laccase enzyme production, specs, QC, pilot validation, cost-in-use, and supplier qualification for industrial applications.

A practical procurement guide for selecting laccase enzyme suppliers, validating production quality, and scaling use in wastewater treatment, pulp bleaching, lignin modification, and bioremediation.

What Industrial Buyers Mean by Laccase Enzyme Production
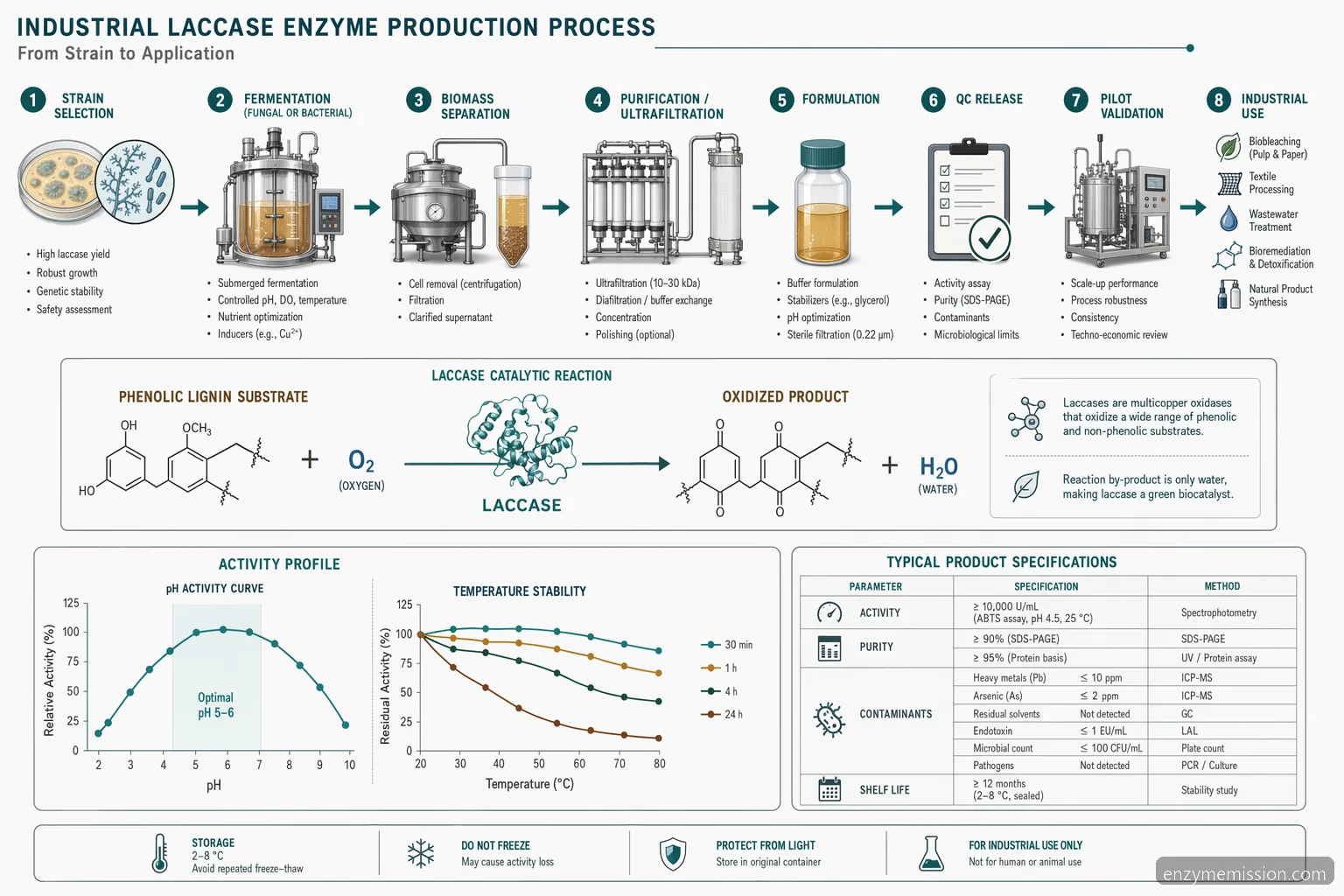
For B2B procurement, laccase enzyme production refers to the complete manufacturing route used to deliver a consistent oxidoreductase product at industrial scale. It includes strain selection, fermentation, downstream separation, formulation, packaging, and release testing. The buyer’s concern is not only activity per gram or milliliter, but whether that activity performs under real plant conditions such as variable wastewater composition, pulp furnish, lignin content, salts, residual peroxide, or process temperature. A qualified supplier should explain the production of laccase enzyme in terms relevant to your application: source organism, unit definition, stabilizers, recommended storage, shelf life, and compatibility limits. For industrial use, laccase is commonly supplied as liquid concentrate or dry powder, with each format affecting dosing accuracy, logistics, and storage. The best purchasing decision comes from matching enzyme specification to process economics rather than choosing the highest activity number alone.
Confirm the unit definition used for enzyme activity. • Request application-specific guidance, not only catalog data. • Evaluate liquid versus powder based on handling and storage.
Fungal and Bacterial Production Routes
Laccase enzyme production from fungi is common because many white-rot and filamentous fungi secrete extracellular laccase into the fermentation broth, simplifying recovery compared with intracellular products. Production and purification of laccase enzyme through fungi typically involves submerged or solid-state fermentation, controlled aeration, pH management, biomass removal, filtration, concentration, and formulation. Typical fermentation conditions depend on strain and feedstock, but many fungal processes operate in mildly acidic ranges around pH 4.0 to 6.0 and temperatures near 25 to 32 °C. Laccase enzyme production from bacteria may be selected when the target process needs broader pH tolerance, thermal robustness, or different substrate specificity. Bacterial systems may require different downstream steps depending on whether the enzyme is extracellular, periplasmic, or cell-associated. Buyers should avoid assuming one route is universally better; the practical question is which production route gives reliable activity, purity, stability, and cost-in-use for the intended industrial process.
Fungal routes are often favored for extracellular enzyme recovery. • Bacterial routes can be useful for selected stability requirements. • Ask whether production is submerged, solid-state, or another controlled method.
Purification, Formulation, and QC Release
After fermentation, industrial laccase enzyme production usually moves through clarification, microfiltration or centrifugation, concentration, buffer adjustment, and stabilization. High-purity laboratory enzyme is not always necessary for wastewater treatment, pulp bleaching, or lignin modification; however, uncontrolled impurities can affect color, odor, foaming, viscosity, metal content, or downstream compatibility. A supplier should provide a Certificate of Analysis for each batch and a Technical Data Sheet that states activity, appearance, pH range, temperature range, storage conditions, and recommended handling. The Safety Data Sheet should cover workplace exposure, spill response, and transport information. Common QC checks include activity assay, protein concentration or solids, pH, moisture for powders, microbial limits where relevant, heavy metals if required by the buyer, and stability after accelerated or real-time storage. For production and industrial applications of laccase enzyme, batch consistency and application performance often matter more than a highly purified analytical profile.
Request COA, TDS, and SDS before pilot approval. • Check activity retention after storage and shipping simulation. • Match purity level to process need and budget.
Application Conditions for Laccase Wastewater and Lignin Use
Laccase enzyme performance depends strongly on pH, temperature, oxygen availability, substrate type, inhibitors, and contact time. In many wastewater treatment trials, screening starts around pH 4.5 to 7.0, 25 to 45 °C, and dosages from 10 to 200 enzyme units per liter, then narrows based on color removal, phenolic reduction, COD impact, toxicity reduction, and sludge behavior. For pulp bleaching or laccase lignin modification, trials often assess pH 4.0 to 6.5, 35 to 55 °C, enzyme charge per oven-dry pulp or lignin solids, and whether a mediator is required. Residual peroxide, sulfites, high salt, heavy metals, surfactants, and extreme pH can reduce performance. Because laccase uses molecular oxygen as the electron acceptor, mixing and aeration can influence conversion. Suppliers should help design a pilot matrix that reflects the plant’s actual feed variability rather than ideal laboratory substrate conditions.
Start with a dosage range, then optimize by response data. • Verify oxygen transfer and mixing during scale-up. • Screen inhibitors present in the real process stream.
Pilot Validation and Cost-in-Use
A buyer-focused laccase enzyme production discussion should lead quickly to pilot validation. The goal is to quantify cost-in-use, not simply purchase price per kilogram. A structured pilot should compare at least three enzyme dosages, two contact times, and relevant pH or temperature set points against a plant baseline. For wastewater, monitor color, phenolics, COD, BOD, toxicity indicators if required, sludge volume, dewatering behavior, and any effect on downstream biological treatment. For pulp bleaching, monitor brightness, kappa number, viscosity, chemical reduction, effluent load, and pulp strength. For bioremediation, track target compound degradation and by-product formation. Cost-in-use includes enzyme dose, dilution water, pH adjustment, aeration, mediators if used, retention time, tank capacity, labor, storage losses, and waste handling. A reliable supplier should support bench screening, pilot trial design, performance review, and scale-up assumptions before commercial supply is committed.
Compare enzyme cost against total process savings. • Use real process material, not only synthetic substrates. • Define success criteria before starting the pilot.
Supplier Qualification for Laccase Enzyme Production
Supplier qualification should confirm that the manufacturer can deliver consistent laccase enzyme at the required scale, documentation level, and lead time. Ask for recent COA examples, TDS, SDS, standard packaging options, storage recommendations, allergen or sensitizer handling statements where applicable, and change-control practices for strain, process, raw materials, or formulation. Buyers who request a laccase enzyme production pdf or laccase enzyme production ppt are usually looking for technical transparency; the useful version should include process overview, specifications, QC methods, application data, and pilot recommendations without disclosing proprietary know-how. Evaluate whether the supplier can provide retained samples, batch traceability, pre-shipment samples, and support for import documentation. Avoid relying on generic activity claims without method details. A qualified supplier should be able to discuss production of laccase enzyme, application risks, storage stability, and practical troubleshooting in a way that supports commercial operations.
Require batch traceability and clear change notification. • Review technical documentation before price negotiation. • Confirm lead time, packaging, and minimum order quantity.
Technical Buying Checklist
Buyer Questions
There is no single best source for every industrial process. Laccase enzyme production from fungi is widely used because many fungal strains secrete extracellular enzyme, which can simplify recovery. Bacterial production may be attractive when the process needs different stability or pH performance. Buyers should compare application data, batch consistency, activity method, storage stability, and cost-in-use rather than selecting by organism alone.
Ask the supplier to explain the fermentation type, downstream clarification, concentration, formulation, and QC release strategy at a practical level. You do not need proprietary strain details, but you should receive activity definition, batch COA, TDS, SDS, pH and temperature guidance, storage conditions, and evidence of consistency. For industrial use, the right purification level is the one that performs reliably without unnecessary cost.
A safe starting point for screening is often 10 to 200 U/L, but the correct dosage depends on wastewater composition, phenolic load, color bodies, pH, temperature, inhibitors, oxygen transfer, and target outcome. Bench trials should test multiple dosages and contact times using actual plant wastewater. Final dosing should be based on performance targets and cost-in-use, not supplier dosage claims alone.
At minimum, request a current Technical Data Sheet, Safety Data Sheet, and batch-specific Certificate of Analysis. For supplier qualification, also ask for storage and shelf-life guidance, packaging options, activity assay method, recommended application conditions, and change-control practices. If you need a laccase enzyme production pdf or ppt for internal review, it should summarize specifications, QC, pilot recommendations, and handling requirements.
Cost-in-use includes the delivered enzyme price plus dose rate, dilution, pH adjustment, aeration, mediators if needed, contact time, tank capacity, storage losses, labor, and downstream effects. A higher-activity product is not automatically cheaper if it is unstable or needs extra conditioning. Run a pilot with real process material and compare total operating cost against the performance benefit.
Related Search Themes
production of laccase enzyme, laccase enzyme production from bacteria, laccase enzyme, production and purification of laccase enzyme through fungi, production and industrial applications of laccase enzyme, laccase enzyme production ppt
Laccase for Research & Industry
Need Laccase for your lab or production process?
ISO 9001 certified · Food-grade & research-grade · Ships to 80+ countries
Frequently Asked Questions
What is the best source for laccase enzyme production?
There is no single best source for every industrial process. Laccase enzyme production from fungi is widely used because many fungal strains secrete extracellular enzyme, which can simplify recovery. Bacterial production may be attractive when the process needs different stability or pH performance. Buyers should compare application data, batch consistency, activity method, storage stability, and cost-in-use rather than selecting by organism alone.
How should I evaluate production and purification of laccase enzyme through fungi?
Ask the supplier to explain the fermentation type, downstream clarification, concentration, formulation, and QC release strategy at a practical level. You do not need proprietary strain details, but you should receive activity definition, batch COA, TDS, SDS, pH and temperature guidance, storage conditions, and evidence of consistency. For industrial use, the right purification level is the one that performs reliably without unnecessary cost.
What dosage should be used for laccase wastewater treatment?
A safe starting point for screening is often 10 to 200 U/L, but the correct dosage depends on wastewater composition, phenolic load, color bodies, pH, temperature, inhibitors, oxygen transfer, and target outcome. Bench trials should test multiple dosages and contact times using actual plant wastewater. Final dosing should be based on performance targets and cost-in-use, not supplier dosage claims alone.
What documents should a laccase enzyme supplier provide?
At minimum, request a current Technical Data Sheet, Safety Data Sheet, and batch-specific Certificate of Analysis. For supplier qualification, also ask for storage and shelf-life guidance, packaging options, activity assay method, recommended application conditions, and change-control practices. If you need a laccase enzyme production pdf or ppt for internal review, it should summarize specifications, QC, pilot recommendations, and handling requirements.
How is cost-in-use calculated for laccase enzyme production purchases?
Cost-in-use includes the delivered enzyme price plus dose rate, dilution, pH adjustment, aeration, mediators if needed, contact time, tank capacity, storage losses, labor, and downstream effects. A higher-activity product is not automatically cheaper if it is unstable or needs extra conditioning. Run a pilot with real process material and compare total operating cost against the performance benefit.
Related: Laccase for Industrial Oxidation Jobs
Turn This Guide Into a Supplier Brief Request laccase samples, COA/TDS/SDS, and pilot support for your industrial process. See our application page for Laccase for Industrial Oxidation Jobs at /applications/laccase-enzyme-price-india/ for specs, MOQ, and a free 50 g sample.
Contact Us to Contribute